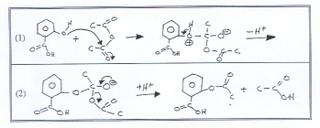
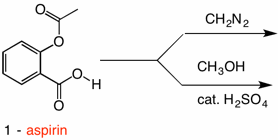
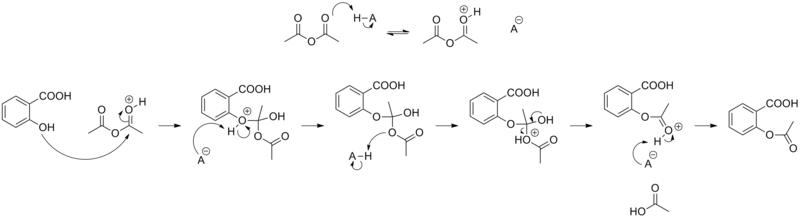
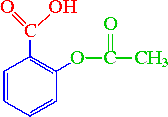
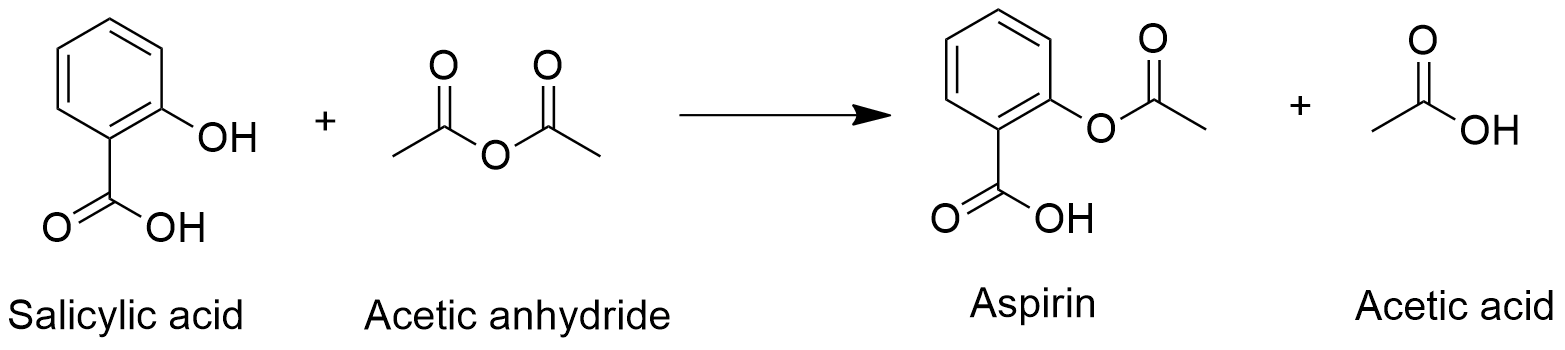
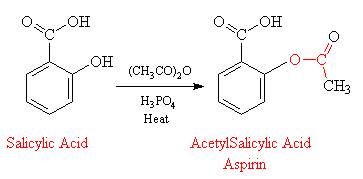
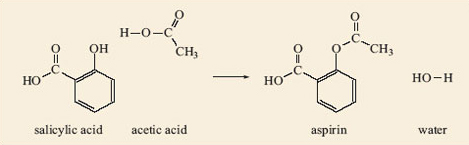
Acid catalysed hydrolysis of aspirins and base catalysed hydrolysis of aspirins which yields more product? - Quora
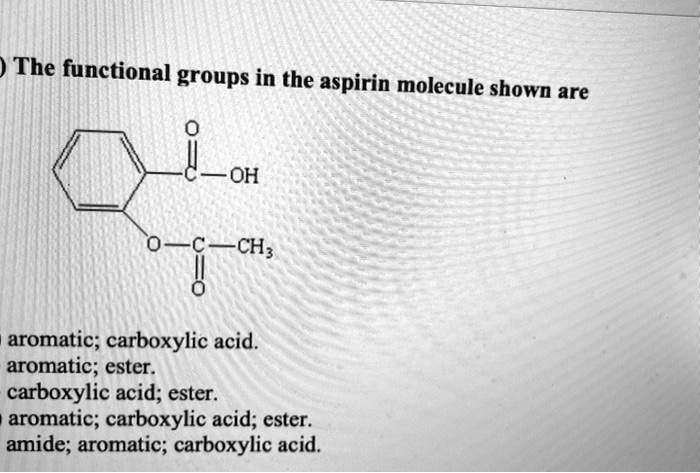
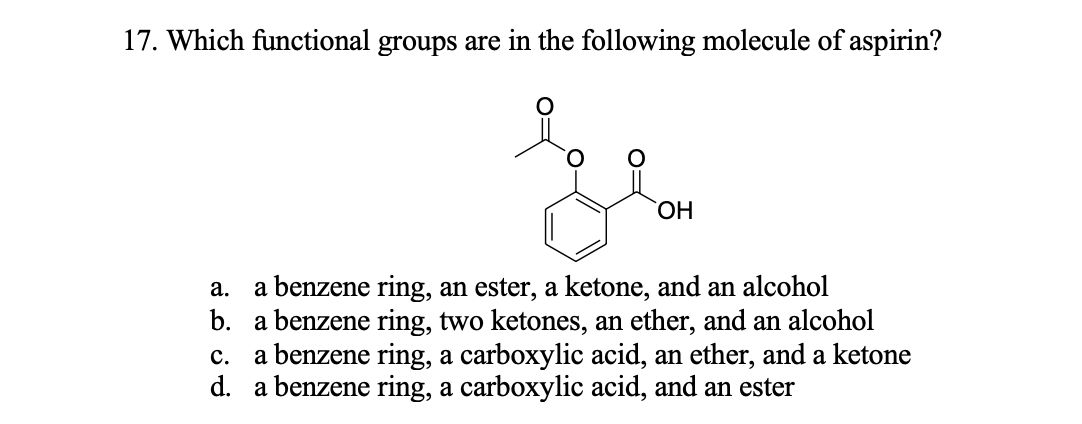
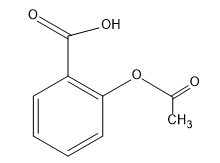
SOLVED: The functional groups in the aspirin molecule shown are OH CH; aromatic; carboxylic acid. aromatic; ester. carboxylic acid; ester: aromatic; carboxylic acid; ester. amide; aromatic; carboxylic acid.

True or false? Challenges and recent highlights in the development of aspirin prodrugs - ScienceDirect
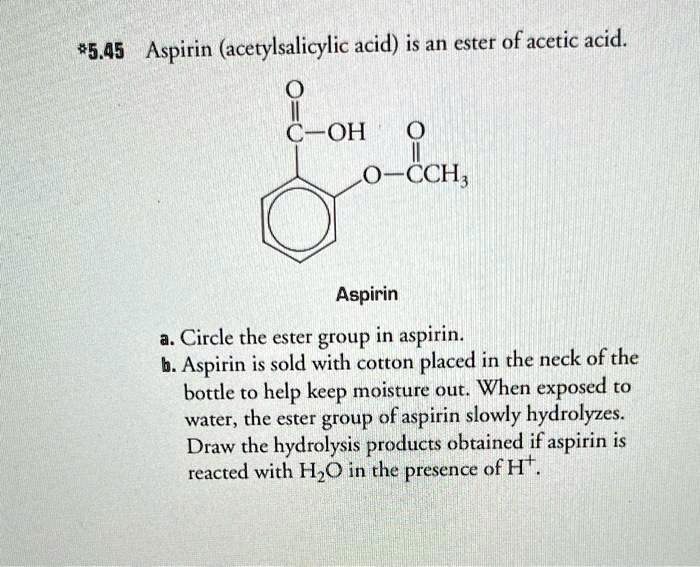
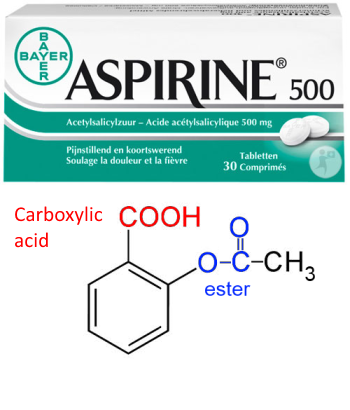
SOLVED: 5.45 Aspirin (acetylsalicylic acid) is an ester of acetic acid (COH O O-CCH). Aspirin is sold with cotton placed in the neck of the bottle to help keep moisture out. When

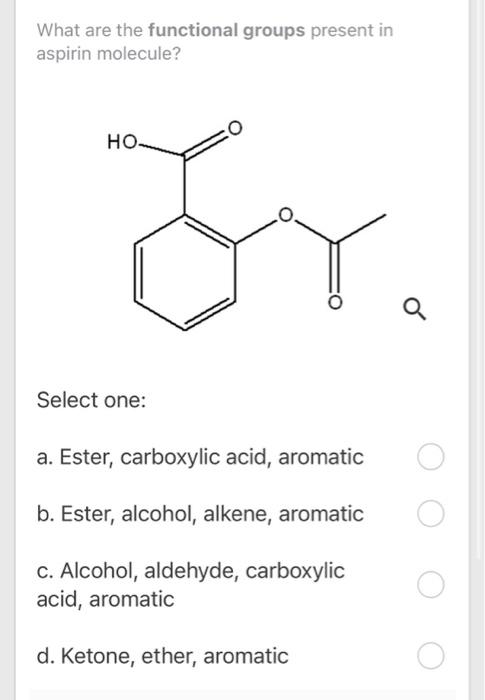
Circle and label the ester group and the carboxylic acid group on the following aspirin structure. | Homework.Study.com