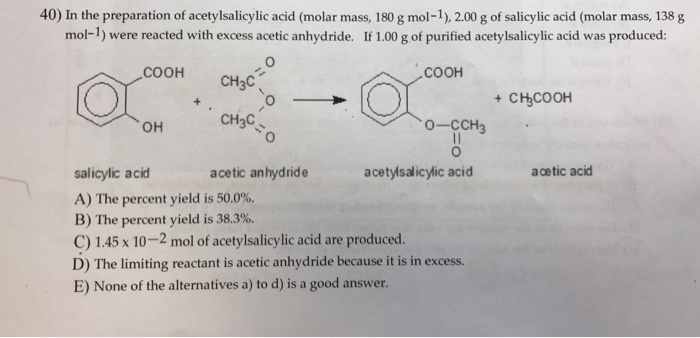
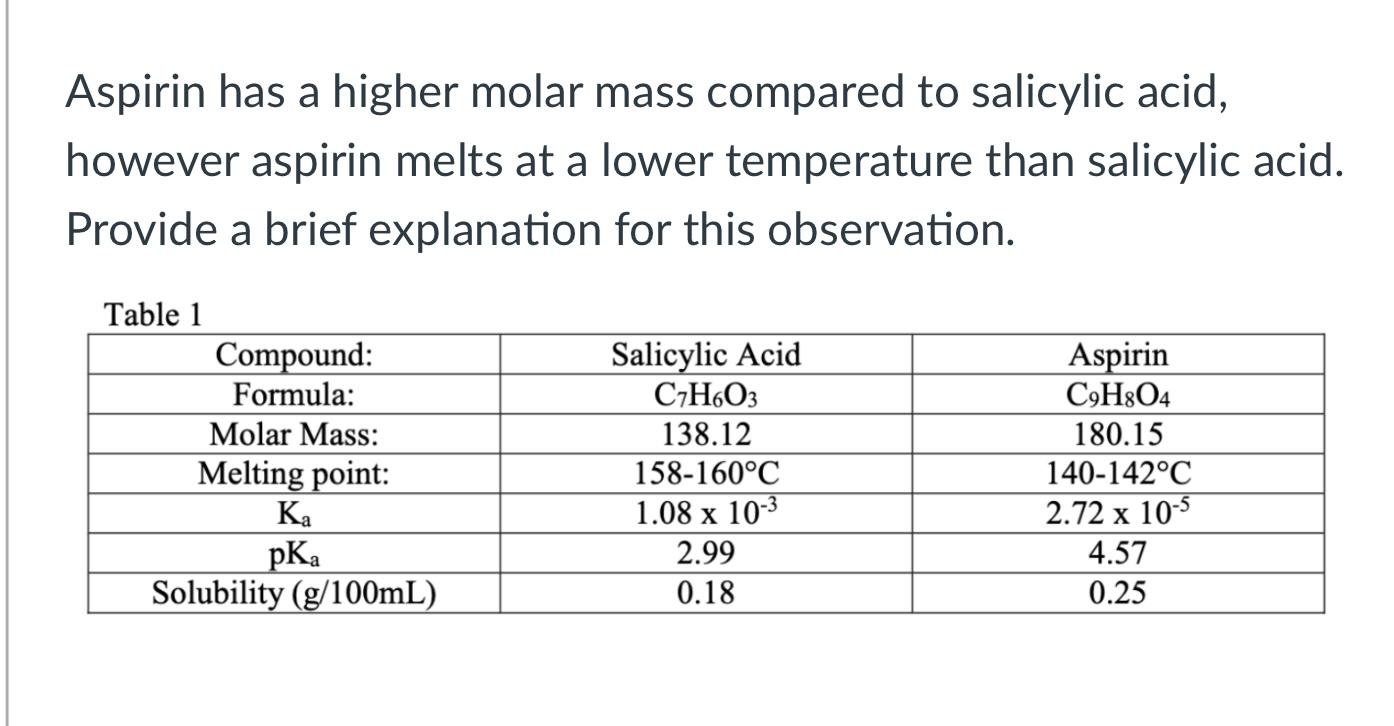
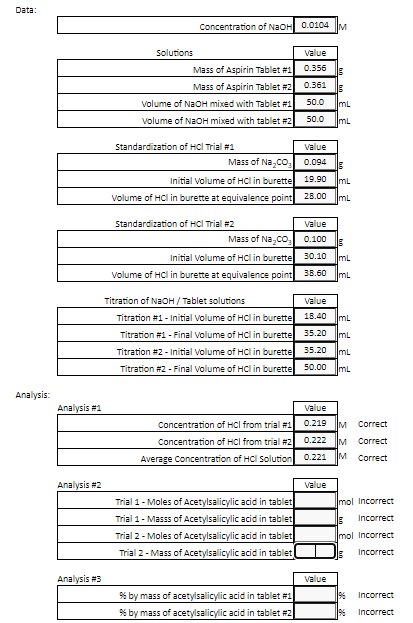
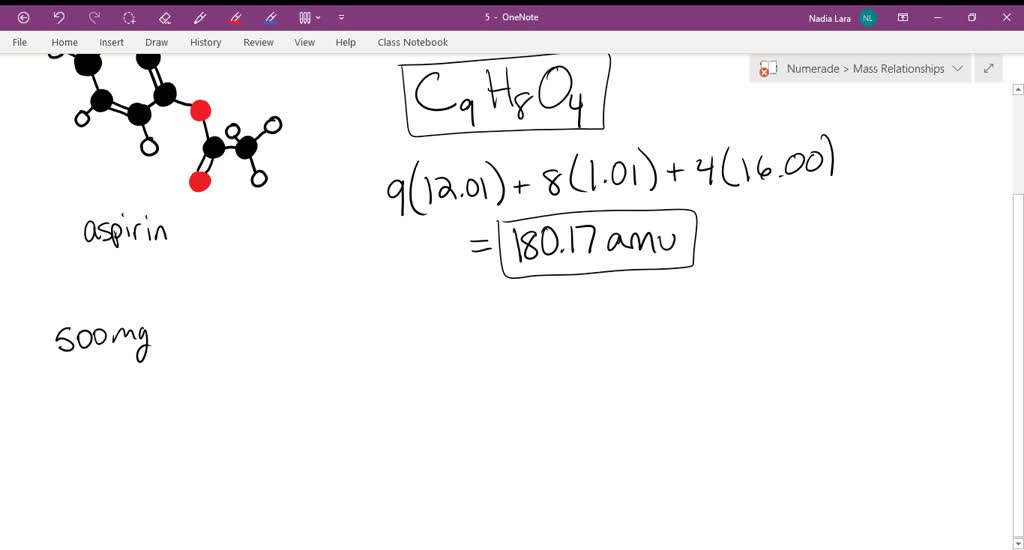
The molecular formula of acetylsalicylic acid aspirin, one of the most commonly used pain reliever - YouTube

Aspirin (acetyl salicyclic acid, molar mass = 180 mol) used as analgesic has pK value of 2. Two tablets of aspirin each weighing 90 mg are dissolved in 100 mL of water.

Sketch the molecular structure of acetylsalicylic acid and calculate its molar mass. | Homework.Study.com
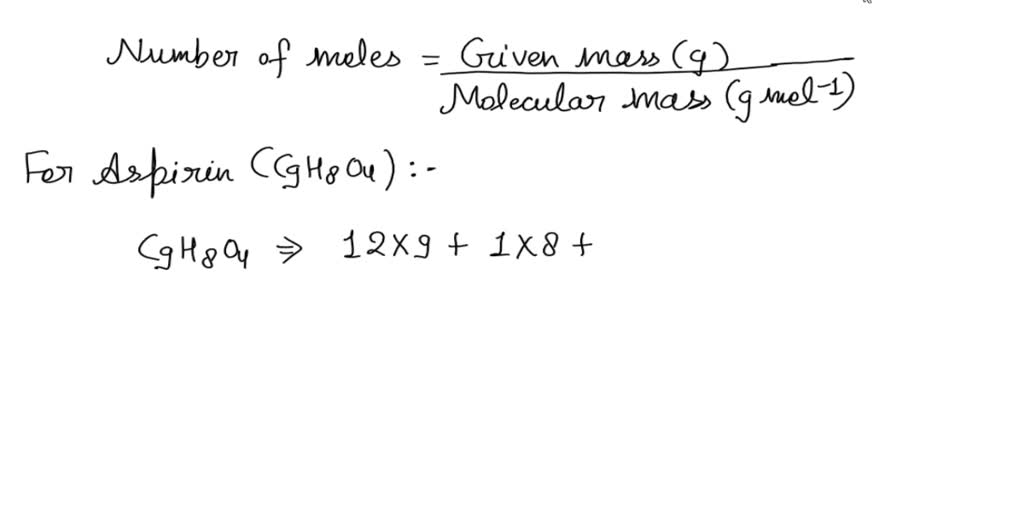
SOLVED: What is the molecular weight of aspirin (C9H8O4)? 180.157 amu What is the mass of 0.295 mol of aspirin? How many moles of aspirin are present in 400 mg of aspirin?

Calculate the mass percentage of aspirin (C9H8O4) in acetonitrile (CH3CN) when 6.5g of C9H8O4.... - YouTube
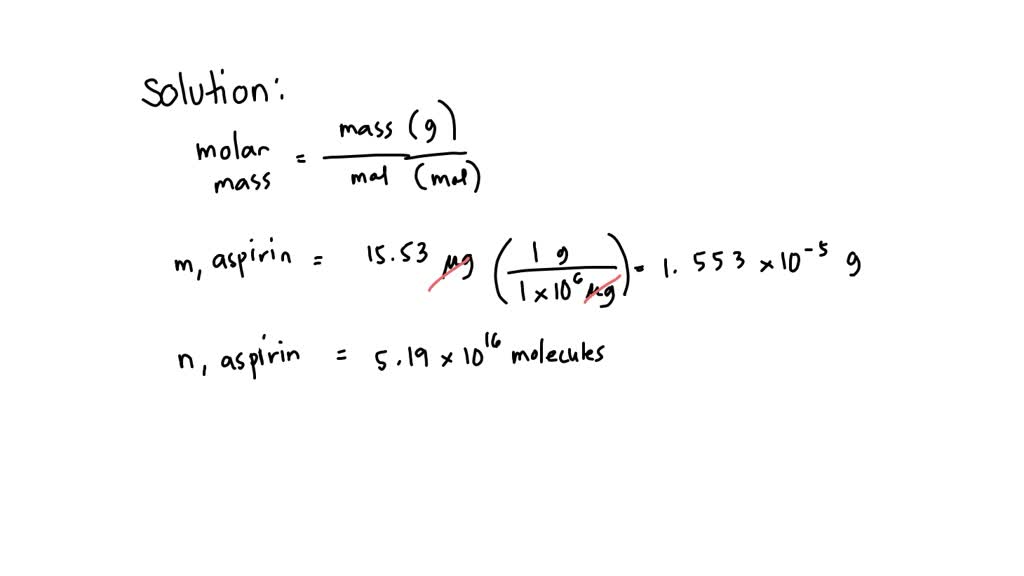
SOLVED: What is the molar mass of aspirin if 5.19 x 10^16 molecules of aspirin weigh 15.53 g? A) 180 g/mol B) 80.6 g/mol C) 133.8 g/mol D) 200 g/mol

Chemical structure and molecular weight (m.w.) of traditional aspirin,... | Download Scientific Diagram
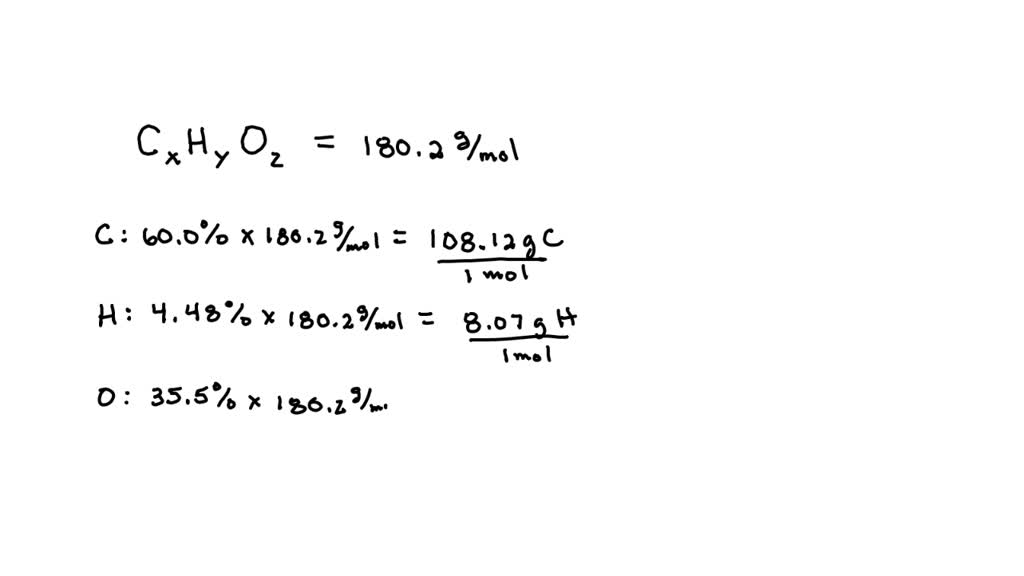
SOLVED: Aspirin has a molar mass of 180.2 g/mol and a composition of 60.0 wt% C, 4.48 wt% H and 35.5 wt% O. What is a molecular formula for aspirin?